While UC San Diego has adapted to the ever-changing COVID-19 situation and has made plans to lift many of its mandatory masking guidelines by Dec. 16, scientists are still learning about the lasting impact on those who have already contracted the virus. Despite the numerous known diverse neurological symptoms associated with long COVID, the pathology of these debilitating conditions have remained unclear. Researchers at the UCSD School of Medicine and Sanford Consortium led an international team of researchers to investigate the effect of SARS-CoV-2 on brain cortical organoids.
Post-COVID conditions, or more commonly known as long COVID, is defined by the Center for Disease Control and Prevention as the phenomenon where individuals “infected with the virus that causes COVID-19 … experience long-term effects from their infection.” Some of the long-term neurological effects include difficulty concentrating or thinking, loss of smell or taste, depression, and anxiety.
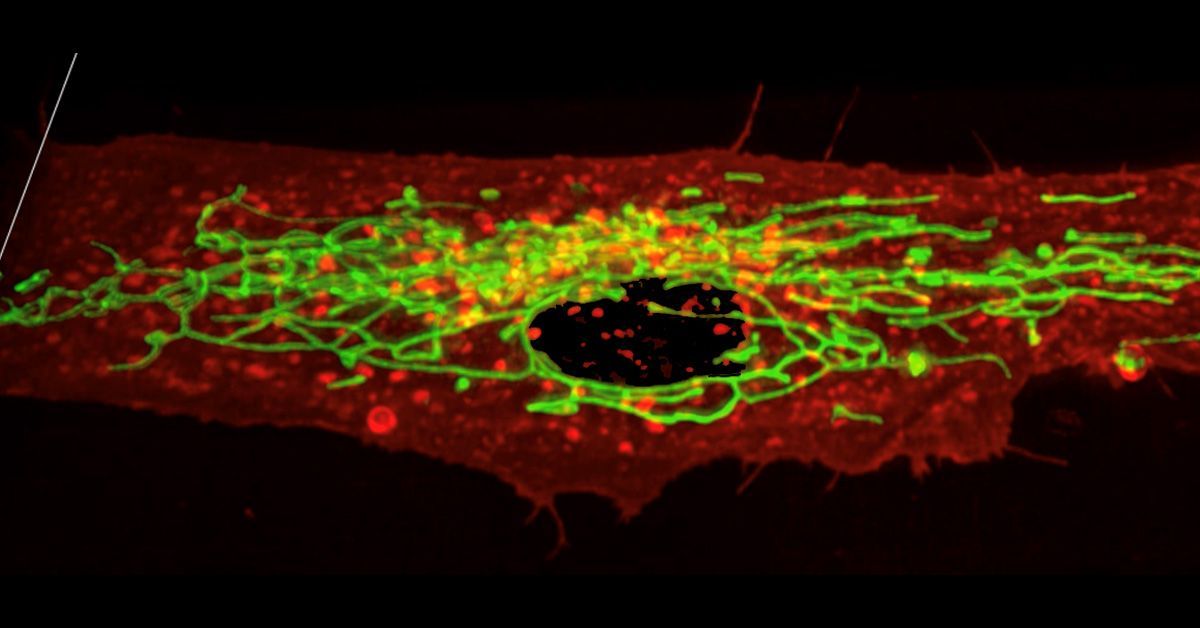
SARS-CoV-2, the virus that causes COVID-19, is not only able to infect brain organoids, but also proliferate within them. Specifically, researchers have found that SARS-CoV-2 viral levels peak at 48 hours post-infection, before declining over the remaining course of infection. This SARS-CoV-2 infection and replication in brain organoids is additionally associated with a loss of excitatory synapses in the neurons, and results in neuronal cell death.
After establishing that SARS-CoV-2 promotes neuronal death in brain organoids, the researchers were curious if Sofosbuvir, a U.S. Food and Drug Administration-approved antiviral drug that is already used to treat Hepatitis C, would be able to mitigate some of these effects. Sofosbuvir has previously been shown to suppress other structurally similar viruses, such as coronaviridae.
The researchers found that Sofosbuvir is able to inhibit SARS-CoV-2 replication. Furthermore, they identified an appropriate dose of Sofosbuvir that would allow for the maximum inhibition of SARS-CoV-2 without inducing any cell death.
In a press release, senior study author and Professor in the department of pediatrics, and cellular and molecular medicine, at the UC San Diego School of Medicine Alysson R. Muotri spoke about the significance of his team’s results in the clinical landscape of COVID-19.
“Vaccines and emerging treatments have reduced the health consequences of COVID-19 in most patients,” Muotri explained. “But the phenomenon of Long COVID, characterized by persisting symptoms that include neurological impairment, remains poorly understood and without any specific remedy.”
He continued to explain how their findings on Sofosbuvir potentially has far-reaching impacts for patient outcomes.
“The bottom line is that Sofosbuvir appears to have the potential to arrest or prevent the development of neurological symptoms in COVID-19 patients,” Muotri said. “And because it has been shown to present no safety concerns in pregnant women, it might also be an option for preventing SARS-CoV-2 transmission to their unborn children.”
However, Muotri notes that much work still needs to be done, in order for her team’s findings to be safely applied in a clinical setting.
“Further studies and clinical trials are needed, of course, but these findings offer a path forward for treating a condition (Long COVID) that has so far stymied remedy for millions of people worldwide,” Muotri said.
The researchers’ published findings can be found in PLOS Biology.
Photo by Alexander Olsen for the UCSD Guardian.
Correction: This article was updated on Dec. 1 at 6:06 pm to correct the mentioned researcher’s pronouns.




















joseph mcfadden • Nov 21, 2022 at 4:12 am
this kind of stuff is maddening … infuriating. so let me get this straight;
you already HAVE a drug that the “U.S. Food and Drug Administration-approved antiviral drug that is already used to treat Hepatitis C” – so its already been used and tested and approved.
AND you have seen excellent results in long-covid patients. AND its proven safe.
YET — “much work still needs to be done, in order for her team’s findings to be safely applied in a clinical setting.” — i’m sorry but wasn’t that work already done?
so now the “…stymied remedy for millions of people worldwide” is still …. going no where?!
why? – how about an article on THAT.
but hey, at least you got published in a journal huh. nice. that sure does a lot for my long covid. thanks!